Results of sampling assignment for potential salmonella in lettuce revealed
- Like
- Digg
- Del
- Tumblr
- VKontakte
- Buffer
- Love This
- Odnoklassniki
- Meneame
- Blogger
- Amazon
- Yahoo Mail
- Gmail
- AOL
- Newsvine
- HackerNews
- Evernote
- MySpace
- Mail.ru
- Viadeo
- Line
- Comments
- Yummly
- SMS
- Viber
- Telegram
- Subscribe
- Skype
- Facebook Messenger
- Kakao
- LiveJournal
- Yammer
- Edgar
- Fintel
- Mix
- Instapaper
- Copy Link
Posted: 16 August 2019 | Rachael Harper (New Food Magazine) | No comments yet
The FDA tested romaine lettuce samples grown in the Yuma region after the E.coli outbreak in 2018 which killed five people.
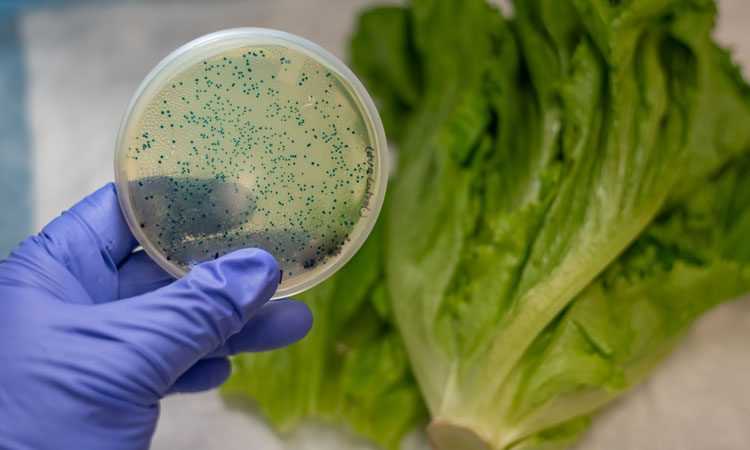

The results of a sampling assignment that tested romaine lettuce grown in the Yuma, AZ agricultural region for pathogenic Shiga toxin-producing Escherichia coli (STEC) and salmonella spp has been released by the US Food and Drug Administration (FDA).
This was conducted following the spring 2018 outbreak of E.coli which was linked to romaine lettuce from the Yuma growing region. The outbreak involved 210 people in 36 states and resulted in 96 hospitalisations, 27 cases of hemolytic uremic syndrome (a type of kidney failure) and five deaths.
In addition, there have been five suspected or confirmed multistate outbreaks of foodborne illness linked to produce harvested in the Yuma growing region since 2012.
The findings of this assignment suggest that there was no widespread STEC or salmonella spp. contamination of romaine lettuce from the Yuma growing region during the period when the sampling occurred.
The FDA began sampling romaine lettuce in December 2018, shortly after the season’s harvest began, to monitor the risk of contaminated romaine lettuce entering the market from this region. It chose to sample romaine lettuce at commercial coolers and cold storage facilities where field heat is removed from harvested romaine and product is cold-stored before shipment.

The FDA testing samples of romaine lettuce grown in the Yuma for STEC and salmonella.
This enabled the agency to collect samples from multiple farms at the same time from centralised locations, while also being able to easily identify the farms from which each sample originated. In all, FDA field staff visited 26 commercial coolers and cold storage facilities in the Yuma growing region with the assistance of the Arizona Department of Agriculture and the Arizona Department of Health Services.
During the assignment, the FDA collected and tested 118 samples of romaine lettuce for each pathogen. The FDA did not detect salmonella in any of the samples. The agency detected Shiga toxin-producing E.coli (STEC) in a single sample, however, further analysis determined that the bacteria was not pathogenic.
The FDA has said it is continuing to work with leafy greens stakeholders in the Yuma region to consider a longer-term environmental study to identify and control risks that will prevent future outbreaks, with the ultimate goal of protecting consumers.
Related topics
Contaminants, Food Safety, Regulation & Legislation, The consumer







