Food sector regulation: preventing coronavirus 2.0
- Like
- Digg
- Del
- Tumblr
- VKontakte
- Buffer
- Love This
- Odnoklassniki
- Meneame
- Blogger
- Amazon
- Yahoo Mail
- Gmail
- AOL
- Newsvine
- HackerNews
- Evernote
- MySpace
- Mail.ru
- Viadeo
- Line
- Comments
- Yummly
- SMS
- Viber
- Telegram
- Subscribe
- Skype
- Facebook Messenger
- Kakao
- LiveJournal
- Yammer
- Edgar
- Fintel
- Mix
- Instapaper
- Copy Link
Posted: 25 March 2020 | Rob Tymowski | No comments yet
Rob Tymowski, Managing Director at Livingstone, provides a primer of the food industry’s regulatory agencies and related frameworks, and how their future of regulation could be essential in preventing another pandemic.


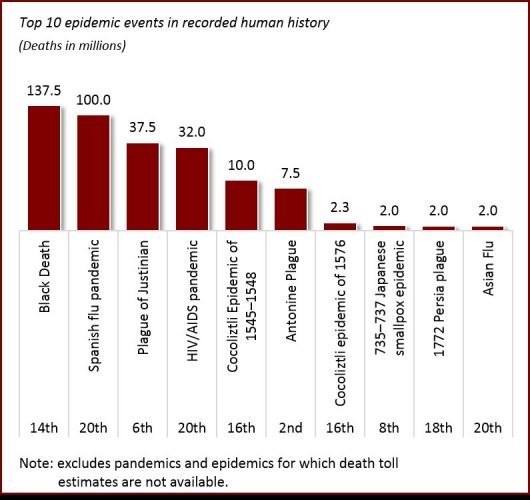
Since the beginning of recorded history, deadly bacterial and viral agents have plagued humanity. Wikipedia identifies 231 epidemics from as early as the fifth century BCE. Of these, 97 include estimated death toll figures.
Of course, bacteria were not discovered until the end of the 17th century; while viruses were identified at the end of the 19th century. Further, the relationship between pathogens and disease wasn’t discovered until Louis Pasteur (namesake of pasteurisation) conducted experiments in the 1860s. For these reasons, we can forgive (or at least excuse) our ancestors for the estimated 120 to 285 million people who died as a result of epidemics prior to the 20th century (note these death toll figures exclude epidemics for which no records exist and/or no reasonably accurate death toll estimate is available). Lack of understanding as the core causes of infectious diseases made our ancestors practically powerless to stop these epidemics. Impacted populations could only pray and wait for these pestilences to run their course.

With the exception of HIV/AIDs, COVID-19 is on pace to become one of the deadliest epidemics since 1900.
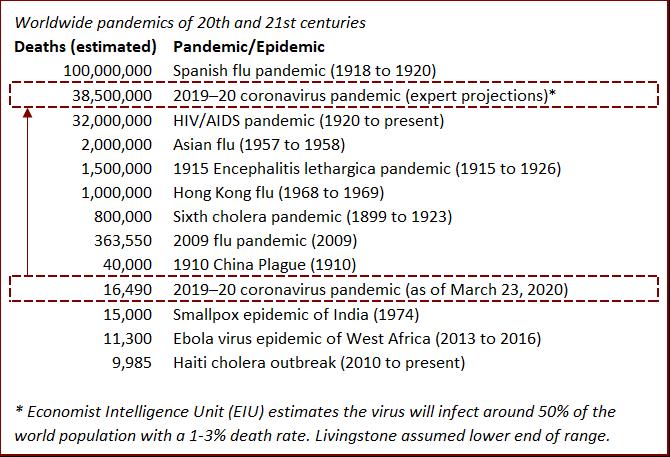
Since the beginning of the 20th century, over 138 million people have died of epidemics and pandemics on a global basis. More accurate death toll numbers exist for this period in comparison to previous centuries. The majority of these deaths were caused by 14 worldwide pandemics, including: the Spanish Flu (up to 100 million | 1918 to 1920); HIV/AIDS (32,000,000 | 1920 to present); the Asian Flu (2 million | 1957 to 1958); the Hong Kong Flu (1 million | 1968 to 1969); and the 2009 Flu Pandemic (575,400 | 2009).
As of 23 March 2020, COVID-19 (commonly known as the coronavirus) ranks tenth on the death toll list since 1900, and has claimed approximately 16,500 lives. Considering this historical context (and with the exception of the on-going HIV/AIDs pandemic), modern man has been remarkably successful in limiting the impact of epidemics, especially since the 1970s and particularly when considering we have a much higher population today than at any other time in human history. We have our modern understanding of medicine and disease prevention techniques to thank for this success.

Each epidemic has an origin story. These stories often begin with:
- An insect bite (most notoriously from mosquitoes and fleas, which operated with impunity governed only by local environmental conditions for most of human history)
- Live species-to-species transfer (e.g., HIV/AIDs, which jumped from chimpanzees or gorillas to humans sometime in the late 19th or early 20th centuries)
- Consumption of undercooked food (e.g., COVID-19; a twist on the species-to-species transfer, which reportedly derived from the stomach contents of a bat or a pangolin in China’s Hubei province).
Once “patient zero” is infected, the spread of epidemics often also occurs via communicable means, from person to person.
Unfortunately, the recent (and ongoing) pandemic caused by COVID-19 provides an all-too real example of tainted food leading to a deadly worldwide outbreak. With the exception of HIV/AIDs, COVID-19 is on pace to become one of the deadliest epidemics since 1900. The question becomes, “what can we do about it?” Social distancing, quarantining and washing hands – while all important and appropriate answers to this question – are beyond the scope of this thought piece. Instead, we focused on preventing the next epidemic that might otherwise arise from undercooked, unchecked, or otherwise unsafe foods.
Food producers, processors, packagers, shippers, retailers, preparers and, ultimately, consumers are all expected to abide by applicable regulations.
This article provides a primer of the food industry’s regulatory agencies and related frameworks, with a particular focus on North American and European markets. These agencies oversee every facet of food ingredient sourcing, production, packaging, transportation, storage, retail and preparation.
The increasing complexity and cross-border interdependence of these regulations all-but guarantee that the demand for third-party-provided food testing, inspection, certification, and compliance (TICC) services will continue to grow for the foreseeable future. These services are increasingly important in today’s culinary environment.
Governing food safety agencies and standards
Every step of the food supply chain is regulated by one or more governing bodies, depending on the applicable region(s) involved. Food producers, processors, packagers, shippers, retailers, preparers and, ultimately, consumers are all expected to abide by applicable regulations. This complicated mix of parties and regulations evolves continuously, requiring industry specialists to keep up with new standards. A primer-level summary of applicable regulatory agencies and governing standards in North America and Europe follows:
Global (but applicable in the US for both imported and exported food products)
- International Organization for Standardization (ISO) is an independent, non-governmental international organisation with a membership of 164 national standards bodies. ISO’s international standards and related documents, cover almost every industry, from technology, to food safety, to agriculture and healthcare. Most recently issued food-related standard is ISO 22000 – Food Safety Management
- Global Food Safety Initiative (GFSI) was created in 2000 to address global food safety crises and help build consumers’ trust in the food they buy, no matter where the food has come from or where in the world they live. GFSI maintains the widely-accepted FSSC 22000 food safety management system, which certifies organisations for food-related compliance (although GFSI is not recognised by ISO, and ISO is not recognised by GFSI). Many experts view FSSC 22000 as a benchmark for food safety
- International Featured Standards (IFS), which was founded in 2003 to focus on food matters, but today comprises eight different food and non-food standards across the supply chain. IFS does not specify what these processes must look like, but provides a risk-based assessment of each. The IFS’s standards are used by manufacturers and retailers worldwide to meet new requirements for quality, transparency, and efficiency related to globalisation
- Safe Quality Food Institute (SQFI), a division of Food Marketing Institute (FMI), is responsible for writing and maintaining the SQF Program (recognised by GFSI) certification as well as a variety of other supporting resources and industry programs that cover food safety, quality, sourcing, and sustainability.
United States
In addition to global regulatory bodies and their associated standards, at least a dozen US-based federal agencies implement more than 35 statutes to make up the federal part of the food safety system (note: excludes state-level parties and rules). 28 House and Senate committees provide oversight of these statutes. The primary Congressional committees responsible for food safety are the Agriculture Committee and Commerce Committee in the House; the Agriculture, Nutrition, and Forestry Committee and the Labor and Human Resources Committee in the Senate; and the House and Senate Agriculture, Rural Development, and Related Agencies Appropriating Subcommittees.
Four agencies play major roles in carrying out food safety regulatory activities, with more than 50 interagency agreements (not listed here) to tie the activities of the various agencies together:
- Food and Drug Administration (FDA), which is part of the Department of Health and Human Services (DHHS)
- Food Safety and Inspection Service (FSIS) of the US Department of Agriculture (USDA)
- Environmental Protection Agency (EPA)
- National Marine Fisheries Service (NMFS) of the US Department of Commerce (USDC).
Canada
Since 2016, regulatory bodies in the US and Canada have generally recognised each other’s food safety systems as comparable to each other. This was the second time that the FDA recognised a foreign food safety system as comparable, the first being New Zealand in 2012. By recognising each other’s systems, the US and Canada can leverage each other’s regulatory systems. For example, each partner will consider the oversight of the other when prioritising inspection activities. Beyond inspection and admissibility, systems recognition establishes a framework for regulatory cooperation in a variety of areas that range from scientific collaboration to outbreak response, and provide the baseline level of public health protection that helps assure the safety of exported foods from each country. Systems recognition will help the FDA be more risk-based in planning the scope and frequency of its inspection activities, including foreign facility inspections, import field exams, and import sampling.
- Department of Health Canada (Health Canada), which is responsible for establishing standards for the safety and nutritional quality of all foods sold in Canada
- Canadian Food Inspection Agency (CFIA), which is responsible for administration and enforcement of Canada’s many food-related acts and regulations, including the Food and Drugs Act.
European Union / United Kingdom
- European Food Safety Authority (EFSA), which provides scientific and technical support to the European Commission and EU countries in all areas affecting food safety. The EFSA is also responsible for coordinating risk assessments, identifying emerging risks and advising on crisis management
- British Retail Consortium (BRC) focuses on making a positive difference to the retail industry – of which food and other consumable products are subsectors – and the customers it serves, today and in the future. BRC’s mandate includes review of food labeling, food-related emerging risks, and imitation food products
- Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH), which were European Commission environmental regulations first introduced in 2007 to codify the chemical composition and risks associated with existing and new substances, as tracked by the European Chemicals Agency (EHCA) in Helsinki, Finland.
Conclusion
As populations grow, appetites evolve, and global supply chains continue to develop, new food TICC services will be increasingly required to keep consumers safe. Having listed many, though not all, of the applicable governing food safety organisations and standards, one doesn’t have to wonder why many companies in the food industry are turning to third-party professionals to assure compliance both within and across sovereign borders.
To fully appreciate the importance of food standards and the food testing, inspection and certification companies responsible for maintaining them, ask yourself the following questions about the last meal you enjoyed:
- From which country(ies) was my food sourced?
- Who (and which company(ies)) grew, picked, transported, processed, distributed, and prepared the food that I’m eating?
- Did the individuals involved in every step of this process follow proper and accepted food safety protocols?
- How sure am I that the food I’m eating is consistent with its label or menu description?
- Is it possible that any of my food has been contaminated (e.g., heavy metals exposure, foreign objects, biological pathogens, etc.)?
If you trust the safety of the food you ate, and generally know the answers to the above questions then you have food safety standards to thank alongside the food testing, inspection and certification companies that ensure the standards are followed.
Increasingly complicated food industry regulatory trends will continue to provide growth opportunities for TICC sector participants with the institutional competencies, discipline, capabilities, and foresight to take advantage of them. As the global “alphabet soup” of regulatory bodies and standards become more difficult to manage, food industry companies will outsource or supplement their in-house employee teams to assure compliance.
Dedicated TICC service providers are uniquely suited to keep up with the global food industry’s evolving standards, such as the FDA’s proposed rule to establish laboratory accreditation programs for food testing. This potential change would disproportionally impact small, sub-scale lab operators, further supporting the value proposition of third-party service providers. There will surely be additional changes forthcoming related to preventing the “next COVID-19”.
Livingstone recently advised on TICC deals in the workplace safety (Diversified Fall Protection’s acquisition of Peak Fall Protection in October 2019) and materials & food testing (May River Capital’s recapitalisation of NSL Analytical Services in January 2020) spaces.
About the Author
Robert F. Tymowski joined Livingstone in 2013 after beginning his investment banking career in R.W. Baird & Co.’s Business Services Group in Chicago. During his time in investment banking, Rob has worked primarily on sell-side M&A transactions, gaining experience in both Business Services and Industrial sectors. Rob has also gained valuable cross-functional expertise staffed on deals of Livingstone’s Debt Advisory and Special Situations practices. As a senior banking professional, Rob is focused on sourcing and executing Industrial, Commercial, and Residential Services transactions.
Prior to pursuing his MBA, Rob spent four years with Walbridge, a Detroit-based private general contractor and design-builder, where he specialised in industrial, education, and government projects across North America. Rob was a member of Walbridge’s leadership development programme, through which he performed a number of mission-critical roles, including engineer, estimator, scheduler, and project manager.
Rob earned his MBA with emphases in Finance and Accounting, ME in Construction Engineering & Management, and BSE in Civil Engineering, all from the University of Michigan.
Related topics
COVID-19, Food Safety, Outbreaks & product recalls, Regulation & Legislation, Supply chain, The consumer









