UHT processing of milk
- Like
- Digg
- Del
- Tumblr
- VKontakte
- Buffer
- Love This
- Odnoklassniki
- Meneame
- Blogger
- Amazon
- Yahoo Mail
- Gmail
- AOL
- Newsvine
- HackerNews
- Evernote
- MySpace
- Mail.ru
- Viadeo
- Line
- Comments
- Yummly
- SMS
- Viber
- Telegram
- Subscribe
- Skype
- Facebook Messenger
- Kakao
- LiveJournal
- Yammer
- Edgar
- Fintel
- Mix
- Instapaper
- Copy Link
Posted: 4 July 2012 | Hilton Deeth (University of Queensland) | 1 comment
Milk is a highly perishable food so to enable it to be stored and distributed for consumption without spoilage, and without being a health risk through growth of pathogenic bacteria, it is heat treated. The most common type of heat treatment in many parts of the world is pasteurisation, which is performed at a minimum of 72°C for 15 seconds. This is the least heat treatment needed to destroy most pathogenic microorganisms and it also destroys most spoilage organisms. However, a small number of bacteria remain after pasteurisation and packaging, and can grow during storage. Such growth is slow at low temperature and consequently pasteurised milk is always kept refrigerated. Even under refrigeration, pasteurised milk only keeps for about two weeks1.


One way of extending the shelf-life of milk is to heat it at temperatures high enough to destroy almost all microorganisms, and then store it in sealed containers without contamination by bacteria. There are two ways in which this can be carried out: in-container sterilisation and ultrahigh- temperature (UHT) processing. Both produce a ‘commercially sterile’ product which means the milk does not contain microorganisms which can grow under the normal conditions of storage which, in this case, is room temperature2,3.
In-container sterilisation, which uses canning technology, is a batch operation which involves heating the final containers of milk in an autoclave at 110-120°C for 10-20 minutes. By contrast, UHT processing involves heating the milk in a continuous flow system at about 140°C for a very short time – around five seconds.
While the two procedures have the same microbiological effect they have very different chemical effects on the milk constituents. In-container sterilised milk typically has a marked cooked flavour and a slight brownish colour. On the other hand, UHT milk has much less cooked flavour and very little, if any, brown discolouration. The difference in product quality between the two types of sterilised milk was a major driver for the development of UHT processing for producing a shelf-stable product. However, the major driver for its commercialisation and widespread adoption was the development of aseptic packing whereby the sterilised milk is packed into sterile packages and hermetically sealed4.
Microbiological aspects
The main microbiological aim of UHT processing is to inactivate spore-forming bacteria which could grow during storage and cause spoilage. The main targets are Bacillus species, particularly heat-resistant ones such as B. licheniformis and B. subtilus. Geobacillus stearothermophilus is an extremely heat-resistant sporeformer found in milk but because it only grows at temperatures above about 50°C, it does not cause problems in UHT milk unless the milk is severely temperature abused during storage. In comparatively recent times, another extremely heat-resistant sporeformer, B. sporothermodurans, has caused problems in UHT milk; unfortunately, unlike G. stearothermophilus, this organism is mesophilic, that is, it can grow at room temperature5.
The thermal conditions used for UHT processing are designed to give a 9-log reduction in heat-resistant sporeformers. This is equivalent to a bacteriological index (B*) of 1. A UHT process should have a B* of at least 1. In practice, most UHT plants exceed this requirement by a reasonable margin. Many different temperature-time combinations ranging from 130°C for ~30 seconds to 160°C for less than 0.05 seconds could achieve this goal but in reality very long and very short holding times are not commercially practical. The most common UHT temperatures used commercially range from 137 to 145°C6.
The UHT process
The major steps in a UHT process are as follows:
- Preheating, with or without a holding time
- Homogenisation (for indirect systems)
- Heating to sterilisation temperature
- Holding at sterilisation temperature
- Initial cooling
- Homogenisation (alternative position for direct or indirect systems)
- Final cooling
- Aseptic packaging
The preheating stage takes the temperature from ~ 5°C to ~90°C, using the hot milk post-sterilisation as the heating source in tubular or plate heat exchangers. This heat regeneration step is very important for the energy efficiency of the UHT plant. Over 90 per cent of the heat can be regenerated, although this figure varies with the type of plant. In some plants, the milk is held for some time in a holding tube after preheating, e.g., for 60 seconds at ~95°C as in Figure 1. The major reason for this step is to reduce the amount of fouling, or deposit formation, in subsequent heat exchangers although, as noted below, it can also have a major effect on the quality of the final product by inactivating a natural milk enzyme.
The final heating step to the required sterilisation temperature is achieved by one of two major types of heating, the so-called direct and indirect systems. Direct systems heat milk by direct contact with culinary superheated steam while indirect systems employ heat exchangers in which superheated steam heats the milk indirectly through a stainless steel barrier in the form of either a tube or a plate. Direct systems can be either an injection type in which steam is injected into the milk, or an infusion type in which milk is infused into a chamber of superheated steam. The major difference between direct and indirect systems is the rate at which the milk is heated. Direct systems heat milk from preheat temperature to sterilisation temperature in less than one second whereas indirect systems can take several seconds to minutes. The major consequence of this difference is that, for the same bactericidal effect, the direct systems produce much less chemical change in the milk constituents than the indirect systems7.
When the sterilisation temperature is reached, the milk enters a holding tube. The temperature of the milk and the time it takes to pass through this holding tube are the nominal conditions which are usually cited for a UHT process, e.g., 140°C for five seconds. While this is a convenient convention, it does not give a true picture of the heat process to which the product is subjected. Many chemical and microbiological changes occur in the heating step immediately before and in the cooling step immediately after the sterilisation step and hence these sections of the plant should be taken into account in addition to the sterilisation holding tube when considering the extent of these changes.
The initial cooling of the product in direct systems is achieved very rapidly as it is passed through a vacuum chamber which removes the water condensed into the product during the steam heating and in so doing returns the temperature of the product to close to the temperature from which it was heated, usually around 75°C. In the final cooling step in direct systems, and in both cooling steps in indirect systems, the heat from the hot milk is transferred to the cold milk in the preheating/heat regeneration steps.
When fat is present in the product, such as in whole milk, a homogenisation step is included. This is carried out at 60-70°C, either before or after the sterilisation step. If the homogeniser is downstream of the sterilisation step, it must be aseptic as no bacteria can be introduced after sterilisation. This clearly puts a high demand on the plant operators to ensure the homogeniser is aseptic and for this reason, where possible, homogenisation is carried out before sterilisation. However, it has been found that milk processed by a direct heating process has to be homogenised downstream to break up aggregates of protein which form during heating and cause an astringent taste in the milk.
The aseptic packaging step is a crucial one. The product must be transferred after cooling to the final package and the package sealed without introducing even one bacterial cell. In most commercial plants, the product is held in an aseptic tank before it is sent to the aseptic packer. Various packaging types are available but the most common are paperboard and multilayered plastic. The packages are sterilised before being filled, usually with hot hydrogen peroxide followed by hot air to remove residual peroxide.
Changes in milk during UHT processing
It is inevitable that heating a product such as milk at temperatures up to ~140°C will have some effect on its constituents, in addition to the intended bactericidal effects. Furthermore, storage at room temperature for long periods of time (up to 12 months) causes additional effects.
To consumers used to drinking pasteurised milk which differs little in flavour from raw milk, UHT milk often appears to have a cooked or heated flavour. Modern UHT technology minimises the production of this flavour but most consumers can still detect it and it is one reason why many consumers prefer pasteurised milk8. The typical flavour of UHT milk is due a combination of flavours, the chief of which are sulphurous flavours caused by volatile sulphur compounds released from the whey protein, and the proteins in membrane surrounding the milk fat globule9. Other contributors are the aliphatic carbonyl compounds formed during heating and compounds formed in the Maillard reaction. Immediately after manufacture, UHT milk has a strong sulphurous smell and taste due to hydrogen sulphide and other volatile sulphur compounds such as methane thiol. These compounds are markedly reduced in the first week, presumably through oxidation.
The initial step in the Maillard reaction is the reaction between lactose and lysine in milk proteins, chiefly whey proteins. In fact, the extent of this reaction is an indication of the intensity of heat given to the milk. In practice, it is measured as furosine, a product formed when the lactose-containing protein is subjected to acid hydrolysis. Another indicator of the heat treatment is lactulose, an isomer of lactose10.
The whey proteins, particularly β-lactoglobulin which forms about 50 per cent of these soluble proteins in milk, are denatured by heating over about 70°C so that in UHT milk, a large percentage of the whey proteins are in the denatured state and exist largely as complexes with caseins.
The instability of the whey proteins to heat has another consequence during UHT processing. Some whey protein denatures and attaches to the surfaces of the heat exchangers in proteinaceous deposits which obstruct the flow of milk and can eventually cause the plant to be closed down for cleaning. However, this is not the only type of deposit formed during UHT processing. At high temperatures, above about 110°C, calcium phosphate also precipitates on the walls, adding to the ‘fouling’ caused by the whey proteins2.
Surprisingly, the UHT process has only a minimal effect on the nutrient value of milk. There is a small decrease in the water-soluble vitamins but virtually no change in the fatsoluble vitamins. The proteins, in fact, have been shown to be more digestible in UHT milk as a result of the heat treatment11. UHT treatment may also reduce the allergenicity of the milk proteins.
The chemical changes caused by a particular plant can be summarised in a chemical index, C*. A C* of 1 is equivalent to three per cent destruction of the B vitamin, thiamine. UHT plants should be run under conditions which give a C* of less than 1 to avoid excessive chemical damage. Direct heating UHT systems have lower C* values than indirect systems. Therefore a better description of a UHT plant is provided by its B* and C* rather than by the conventionally used temperature-time combination of the sterilisation holding tube.
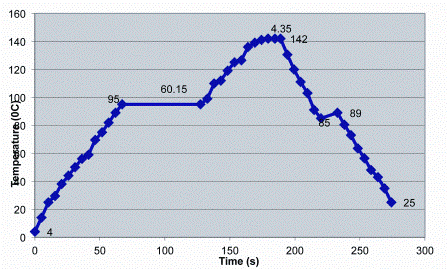

Figure 1: Temperature-time profile of a commercial indirect-heating UHT plant. The nominal holding tube conditions for this plant are 142°C for 4.35 seconds and it’s B* and C* values are 5.77 and 1.52 respectively
Most of the above changes that occur during high-temperature processing of milk have been studied in depth and their reaction kinetics worked out. These enable the changes occurring in a particular UHT plant to be mathematically estimated. The basic information on a plant which allows this to be done is the temperature-time profile. This profile can vary considerably as illustrated in Figures 1 and Figure 2, which show profiles of two commercial UHT installations, an indirect and a direct system, respectively. Therefore when the kinetics for a particular change, e.g., denaturation of β-lactoglobulin, and the temperature-time profile of a UHT plant are known, the effect of that plant on a range of milk components can be predicted. Fortunately, the calculations for doing this can be performed by a computer12 and software for this purpose is available commercially, e.g. NIZO Premia13. The B* and C* values mentioned above can also be computed as they are defined by mathematical formulae. Unfortunately, it is not easy to obtain the temperature-time profile for most plants as a full description of the plant in terms of temperatures and times in all sections is not readily available. Where they are available however, a wealth of information about the overall plant and the product produced it can be obtained. One application of this computer simulation is comparing the effects of small-scale pilot plants with full-sized commercial plants to enable the pilot plants to be configured to match the full-sized commercial UHT equipment6.
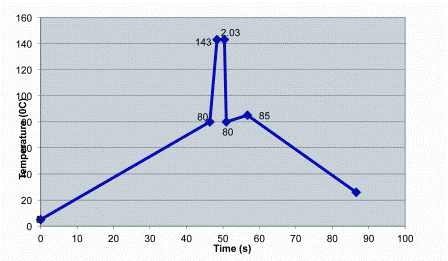

Figure 2: Temperature-time profile of a commercial direct-heating UHT plant. The nominal holding tube conditions for this plant are 143°C for 2.03 seconds, and it’s B* and C* values are 1.26 and 0.17 respectively
Changes in UHT milk during storage
Keeping milk in good condition at room temperature for up to 12 months is a major challenge because of the myriad of changes which can take place. The flavour changes through progress of the Maillard reaction and through oxidation by dissolved oxygen in the milk. The major flavour compounds produced are methyl ketones and aliphatic aldehydes but a large number of flavour compounds are generated. Other flavours which may develop during storage are due to the action of heat-resistant bacterial enzymes which may be present in the raw milk and survive the UHT heat treatment. These include lipases, which break down the fat and form free fatty acids, some of which have strong flavours, and proteases which break down proteins to produce peptides, some of which are bitter.
Another change which can be brought about by proteases is what is known as ‘age gelation’ where the milk thickens over storage and eventually turns into a gel akin to a yogurt14. This undesirable defect can be caused by the heat-resistant bacterial enzymes but it can be also caused by plasmin, a naturally occurring protease in milk, which is quite heat stable and can remain active in UHT milk. Recently, it has been found to be inactivated by some UHT preheating conditions which is an excellent reason for including a holding time in the preheat section of UHT plants15,16.
UHT processing of products other than white milk
UHT processing is now widely used for producing ‘long life’ products such as cream, custard and flavoured milks. However, it is not suitable for making cheese as the curd from UHT milk takes a long time to set and retains a high amount of moisture, giving a very soft and unacceptable cheese. UHT milk is also not very suitable for yogurt manufacture as it forms a very soft gel17; however, it may be more suitable for producing a (long life) drinking yogurt where a firm gel is not required.
References
- Lewis, M. and Heppell, N. 2000. Continuous thermal processing of foods: pasteurization and UHT sterilization, Gaithersburg, Aspen
- Burton, H. 1988. Ultra-high-temperature processing of milk and milk products, London, Elsevier Applied Science
- Deeth, H.C. 2010. Improving UHT techniques. In: M.W. Griffiths (Ed.) Improving the Safety and Quality of Milk. pp. 302-329, Cambridge: Woodhead Publishing
- Robertson, G.L. 2011. Ultra-High Temperature Treatment (UHT): Aseptic Packaging . In: Fuquay JW, Fox PF and McSweeney PLH (eds.), Encyclopedia of Dairy Sciences, Second Edition, vol. 2, pp. 708–713. San Diego: Academic Press
- Pettersson, B., Lembke, F., Hammer, P., Stackebrant, E. and Priest, G. 1996. Bacillus sporothermodurans, a new species producing highly heat-resistant endospores , Int J Systematic Bacteriol, 46, 759–764
- Tran, H., Datta, N., Lewis, M.J. and Deeth, H.C. 2008, Predictions of some product parameters based on the processing conditions of ultra-hightemperature milk plants, Int Dairy J, 18, 939–944
- Datta, N., Elliott, A.J., Perkins, M.L. and Deeth, H.C. 2002. Ultra-High Temperature UHT treatment of milk: Comparison of direct and indirect modes of heating. Aust J Dairy Technol, 57, 211-227
- Perkins, M.L. and Deeth, H.C. 2001. A survey of Australian consumers attitudes towards UHT milk. Aust J Dairy Technol, 56, 28–34
- Al-Attabi, Z., D’Arcy, B.R. and Deeth, H.C. 2009. Volatile sulphur compounds in UHT milk. Critical Reviews in Food Science and Nutrition 491, 28-47
- Elliott, A.J., Datta, N. and Deeth, H.C. 2005. Heatinduced and other chemical changes in commercial UHT milks. J. Dairy Res. 72(4):442-446
- Lacroix, M., Bon, C., Bos, C., Léonil, J., Benamouzig, R., Luengo, C., Fauquant, J., Tomé, D. and Gaudichon, C. 2008. Ultra high temperature treatment, but not pasteurization, affects the postprandial kinetics of milk proteins in humans. Journal of Nutrition 138, 2342–2347
- Browning, E., Lewis, M. and MacDougall, D. 2001. Predicting safety and quality parameters for UHTprocessed milks. Int. J. Dairy Tech., 54, 111–120
- Smit, F., de Jong, P., Straatsma, J. and Verschueren, M. (2001). [NIZO Premia as knowledge management tool for industry] Voedingsmiddelen – technologie, 34, 23–26
- Datta, N. and Deeth, H.C. 2001. Age gelation of UHT milk – a review. Trans Inst Chem Eng C: Food Bioprod Proc, 79, 197-210
- Newstead, D.F., Paterson, G., Anema, S. G., Coker, C.J. and Wewala, A.R. 2006. Plasmin activity in direct-steam-injection UHT-processed reconstituted milk: effects of preheat treatment. Int Dairy J, 16, 573-579
- Van Asselt, A.J., Sweere, A.P.J., Rollema, H.S. and de Jong, P. 2008. Extreme high-temperature treatment of milk with respect to plasmin inactivation, Int Dairy J, 18, 531–538Krasaekoopt, W., Bhandari, B. and Deeth, H.C. 2003. Yogurt from UHT milk: A review. Aust J Dairy Technol. 58, 26-29










hi im hema here. I would like to know about what is pasteurization sterilization and UHT pasteurization. im very confused on it. i though pasteurization, UHT, HTST and sterilization are different.