Nutrifoods 2.0: The impetus towards functional foods and nutraceuticals to face the 21st century
- Like
- Digg
- Del
- Tumblr
- VKontakte
- Buffer
- Love This
- Odnoklassniki
- Meneame
- Blogger
- Amazon
- Yahoo Mail
- Gmail
- AOL
- Newsvine
- HackerNews
- Evernote
- MySpace
- Mail.ru
- Viadeo
- Line
- Comments
- Yummly
- SMS
- Viber
- Telegram
- Subscribe
- Skype
- Facebook Messenger
- Kakao
- LiveJournal
- Yammer
- Edgar
- Fintel
- Mix
- Instapaper
- Copy Link
Posted: 29 October 2014 | Francesc Puiggròs, Ignasi Papell and Lluís Arola, Technology Centre of Nutrition and Health, Universitat Rovira i Virgili | 1 comment
It is widely accepted today that multifactorial aetiologies characterise many chronic diseases, including metabolic syndrome, menopause, and general ageing, and that a lifestyle full of homeostatic disturbances and even shift work can lead to obesity. To combat such diseases and adverse health conditions, an increasing number of functional foods and nutraceuticals have been marketed in recent years as supplements to dietary recommendations. Still, many of these products are marketed according to the promise of health benefits to a single physiological target, rather than more realistically addressing the entire set of factors that may lead to the onset and progression of a multifactorial chronic disease. This article will address innovations in a myriad of new products we believe will become the functional foods of the future, in the process, introducing the new concept of Nutrifoods 2.0.


Multifactorial life conditions in the 21st century
It is not breaking news that the average age, worldwide but most notably in developed countries, has been rising as populations grow older and birth rates continue to decline. But as our society ages, the number of people suffering from chronic non-communicable diseases (NCDs) is also increasing. While the strengthening of primary health care services can deliver long-term care for many disabling illnesses, it is worth asking whether this system really benefits the patients by adding years to their lives rather than life to their years.
NCDs, such as metabolic syndrome, obesity, osteoporosis, diabetes and some chronic neurological diseases, have all been characterised as multifactorial homeostatic alterations of human physiology, as their aetiology includes different but simultaneously occurring risk factors. For example, metabolic syndrome is an assemblage of risk factors, including hypertension, central adiposity, hyperglycaemia and dyslipidaemia that jointly alter an individual body’s homeostasis leading to a high risk of premature cardiovascular disease. The dysregulation of certain adipocytokines that regulate insulin signalling, vascular tone, carbohydrate levels, lipid metabolism and the inflammatory response can contribute to insulin resistance, low grade systemic inflammation and endoplasmatic reticulum stress.
Together, these dysregulations can lead to the development of metabolic syndrome, hypertension, insulin resistance, oxidative stress and vascular maladaptation, in addition to other major contributors to cardiovascular diseases. Osteoporosis is characterised by low bone mass and an increased risk of fracture and is associated with such risk factors as menopause, aging, oxidative stress, genetics and lifestyle. In addition, although Parkinson’s disease aetiology is not completely understood, it seems that its features involve neuroinflammation, oxidative stress, mitochondrial dysfunction and excitotoxicity.
Cluster of compromised physiological targets related to stages in life and contemporary lifestyles
Furthermore, recent nutrition research is now also producing new nutritional strategies to improve upon the stages of an individual’s life. Each life stage, with its specific physiological demands (menopause, childhood, ageing), is typically described by a manifold set of symptoms that often appear at the same time; this results in a clustering effect on the compromised physiological targets. In addition, some modern lifestyles, such as those engaging in shift work or extremely physically demanding occupations (including elite sportsmen), are constantly submitted to jet-lag or challenging environments, which may determine the onset of certain multifactorial, non-communicable diseases. At the very least, these lifestyles can easily compromise one’s own well-being.
For instance, menopause is an expected life stage that often includes hot flushes, increased abdominal fat depots, vaginal dryness, night sweats and moodiness. Alongside the general ageing process, the prevalence of cardiovascular disease risk factors and osteoporosis, hip fractures, falls, sleepiness during the day, fatigue, tiredness and a lack of energy increases.
As an example of the effects of modern lifestyles, it is known that industrialisation has led to over 20 per cent of all industrial workers routinely doing shift work. Much more evidence exists to support the relationship between obesity and certain chronobiological aspects. Shift work can be considered an independent risk factor for the development of obesity and metabolic syndrome. Many epidemiological studies have shown that shift work is associated with a greater prevalence of obesity, hypertriglyceridaemia, low HDL levels, abdominal obesity, diabetes, and cardiovascular disease. Shift workers also demonstrate, as postprandial metabolic responses, increased plasma glucose, insulin, and triglyceride levels, which are associated with impaired melatonin circadian rhythmicity. Although not a disease itself, persistent jet-lag is able to alter the normal functioning of the circadian system, thus linking sleep disorders with obesity. This is why insomnia due to jet-lag and/or shift work is related to obesity and eventually results in significant social problems which require medical care.
Thus, it is important for nutrition researchers to pay more attention to the development of dietary strategies that are able to address each of these life conditions, events and stages that are particularly liable to being assessed in terms of the resultant perceived quality of life.
Dietary vs pharmaceutical strategies
Achieving good population health does not just mean reacting to ill-health, but also proactively preventing the onset of these multifactorial homeostatic alterations by helping people to make healthy choices in terms of both food and lifestyle. The social impact of reducing the morbidity and mortality due to non-communicable diseases (NCDs) is dramatic because 63 per cent of deaths that occur globally are caused by NCDs1. Clearly, modifications of lifestyle and diet are mandatory if we want pharmacological therapies to succeed.
In this respect, metabolic syndrome can be seen as a paradigmatic example of the interactions between nutrition and pharma, two fields that are often merged and possess mutual therapeutic goals. Indeed, combining foods with recognised cholesterol-lowering properties (dietary portfolio) has proven highly effective in lowering the serum cholesterol under metabolically controlled conditions. If one needs to lower his/her cholesterol, new research has revealed that a diet combining soy, nuts, plant sterols, and fibre, known as the portfolio diet, may work better than a traditional low-fat diet. Figure 1 shows that the use of these cholesterol-lowering dietary components in short-term studies that provided all of the food consumed by the subjects was able to reduce serum low-density lipoprotein cholesterol (LDL-C) by 28.6 per cent. This result is very close to the effect achieved by lovastatin, which causes a reduction of 31 per cent2.
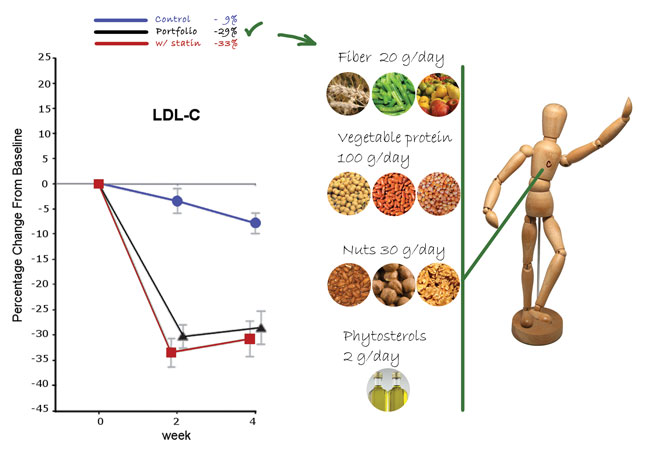

Figure 1: Comparison between the effects of a portfolio diet and those of statin treatment on LDL-cholesterol as an example of combining foods as an effective dietary strategy to face a given risk factor. Adapted from Jenkins et al.2
Currently, most of the NCDs and general health statuses that are compromised by an advancing life stage are being addressed by overused pharmaceutical approaches. The multifaceted realm of nutrition has yet to be fully exploited, and further investigations should be made in order to design optimal treatments for this ever-increasing number of people.
Diet can be considered a potential tool for the prevention of disease that could contribute actively to attaining an increased life span that is characterised by healthy aging. With this in mind, the food industry has found a way to develop new products with value-added differentiations, called functional foods, and nutraceuticals, which provide health benefits beyond their nutritional value and comprise a myriad of health claims, both on their labels and in media ads. Currently, it is very easy find functional products claiming they possess antihypertensive activities, hypocholesterolaemic effects, antioxidant properties, probiotic or prebiotic effects, or regulatory effects on the appetite, among others.
In the last decade, nutrition research has opened up new perspectives on nutrients as signalling molecules that are able to modulate different aspects of the metabolism, thus initiating a new challenge for nutrition and food research. In the view of these possibilities, the concept of optimal nutrition has evolved.
In spite of the huge number of products, the market for functional foods other than vitamins and minerals has certain weaknesses, resulting in the low number of products approved by the European Food Safety Authority.
The most relevant issue to be faced is the lack of scientific data behind the new health claims made on behalf of certain foods. The main reason for this is that few biomarkers of disease prevention potential exist for these NCDs; at present, the scientific community has still not delivered any robust markers. In addition, insufficient study has been performed to develop scientific strategy for assessing research on functional foods and the biological effects of nutraceuticals. Further, certain attempts that have been made have been proven unfeasible due to their being based on pharma concepts rather than designs specific for nutrition.
The future of functional foods will depend on continued advances in nutrition science and the development of innovative technologies by the food industry. It will also depend on our understanding of certain consumer concepts, in particular those related to product applications and the ways in which access to the market place is mediated by the regulatory environment on national, regional and global levels.
The impetus of functional foods; working towards a new era of the Functionals 2.0 concept?
At this point, considering the multifactorial, health-challenging traits implicated in the many conditions and stages of life, a future disruptive evolution in the field of functional foods may emerge. Today, bioactive compounds are responsible for a single functional bioactivity that functional foods may contain. This implies that most of the foods marketed as functional were designed to fight certain multifactorial NCDs by combatting only one of the many risk factors associated with the disease.
Most functional foods target one physiological goal. Related formulation strategies follow the single-ingredient-for-one target or, sometimes, the multi-ingredient-for-one target model. Mixing the biological activities of single ingredients could exert synergistic, complementary or even additive effects for more than one physiological target. Therefore, food companies could adopt a new industrial strategy by taking profit from mixing specific biological effects together to beneficially modulate or reverse isolated homeostatic alterations belonging to a multifactorial situation.
In our opinion, we should counter these industrial research trends towards the creation of a new era of functional foods – Nutrifoods 2.0 (Figure 2) – by uncovering the many effects a single supplement may have across several biological pathways. Thus, if sound data could be produced in corroboration, the multifactorial homeostatic alterations could be improved in a number of situations at once. To this end, technology and nutritional science may alter the research paradigms to identify several bioactive ingredients with differing biological effects on multiple physiological targets within the same NCD or life situation. Therefore, the relationship between the consumption of a given Nutrifood 2.0 and the improvement of human wellness makes sense in modern society.
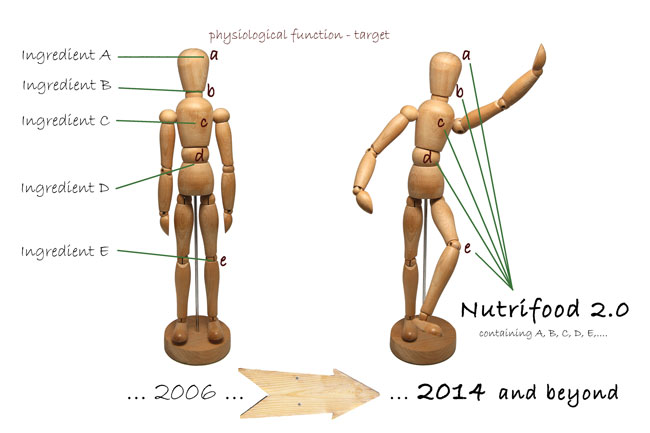

Figure 2: Nutrifoods 2.0 concept. The changing paradigm towards the multi-targeted biological efficacy of innovative functional foods and nutraceuticals
The need for a holistic, mind-body approach
The multifactorial nature of modern NCDs and life situations must be combined with the complex molecular mechanistic interactions underlying their onset in order to progress toward a complete understanding of the overall homeostatic alterations. To uncover novel well-defined biomarkers of multiple risk factors and to elucidate their interactions with a certain bioactive-physiological target until a clear phenotypic effect can be elucidated; we should incorporate ‘foodomic’ technologies into our usual scientific strategies.
Today, ‘foodomics’ is used not only to ensure the presence of nutrient markers in human samples with cutting-edge analytical methods but also to improve the assessment of the biological effects of bioactive compounds in humans using properly developed nutrition trials. This issue has been and remains of key importance for research institutions, food industries, agencies and regulatory laboratories3. In fact, foodomics is intended to be a global discipline that includes all emerging working areas in which food plays a role (including nutrition). Relevant tools include the use of advanced analytical techniques (Figures 3-5) based on traditional GC and LC techniques coupled with MS (GC-MS, LC-MS) and adding 1H NMR, capillary electrophoresis-MS (CE-MS).


Figures 3-5: Tools in foodomics include the use of advanced analytical techniques
Using these techniques, food researchers are able to report any changes in human biofluid metabolomic profiles after the consumption of dietary phytochemicals from a holistic point of view. That is, considering the entire metabolome is more likely thus to prove a real effect. Moreover, the adoption of these tools is unavoidable if we hope to detect the differences between a healthy physiological status and a multifactorial risk factor human status.
For example, in recent years, metagenomics has gained attention due to the use of the gut microbiota as a major topic of research interest in biology. The scientific community is discovering how variations and changes in the composition of the gut microbiota influence normal physiology and contribute to several NCDs, ranging from inflammation to obesity. In fact, more data are called for to propel our knowledge regarding the integration of the microbiota into the gut–brain axis and to uncover whether this modulates its behaviour. Metagenomic studies make possible the holistic study of the relative contributions of the immune, neural and endocrine pathways through which the gut microbiota communicates with the brain. Thus, the role of the gut microbiota may be crucial in a range of gastrointestinal and other disorders, including neuropsychiatric diseases, such as depression and anxiety, and normal brain functioning.
Nutrifoods: The take-home message
From a nutritional view, to effectively improve the health and well-being of an individual who is suffering from multiple risk factors and/or multifactorial-driven life stages, we should focus our innovative activities on a multi-pronged approach that includes the launching of Nutrifoods 2.0 to benefit public health. The items included will become the functional foods and nutraceuticals of the near future, and the food industry should reap the benefits of considering the data from foodomics and new systems biology data, creating Nutrifoods 2.0 using innovative scientific strategies.
References
- World Health Statistics 2011. (n.d.). Retrieved from: http://www.who.int/whosis/whostat/2011/en/
- Jenkins, D. J. A., Kendall, C. W. C., Marchie, A., Faulkner, D. A., Wong, J. M. W., de Souza, R., Connelly, P. W. (2003). Effects of a dietary portfolio of cholesterol-lowering foods vs lovastatin on serum lipids and C-reactive protein. JAMA: the journal of the American Medical Association, 290(4), 502–10. doi:10.1001/jama.290.4.502
- Puiggròs, F., Solà, R., Bladé, C., Salvadó, M.-J., & Arola, L. (2011). Nutritional biomarkers and foodomic methodologies for qualitative and quantitative analysis of bioactive ingredients in dietary intervention studies. Journal of chromatography. A, 1218(42), 7399–414. doi:10.1016/j.chroma.2011.08.051
About the authors
















Enlightening article, educational reach on boundary shattering creative contribution. Wish you all the best for nutraceutical consciousness development.
Dr.Sivam.