UV protection for packaged foods
- Like
- Digg
- Del
- Tumblr
- VKontakte
- Buffer
- Love This
- Odnoklassniki
- Meneame
- Blogger
- Amazon
- Yahoo Mail
- Gmail
- AOL
- Newsvine
- HackerNews
- Evernote
- MySpace
- Mail.ru
- Viadeo
- Line
- Comments
- Yummly
- SMS
- Viber
- Telegram
- Subscribe
- Skype
- Facebook Messenger
- Kakao
- LiveJournal
- Yammer
- Edgar
- Fintel
- Mix
- Instapaper
- Copy Link
Posted: 16 November 2007 | Wolfgang Danzl and Gottfried Ziegleder, Fraunhofer IVV | No comments yet
Transparent plastic packaging is increasingly replacing traditional food packaging made of glass and metal. The advantages of transparent plastic packaging are; its low weight, low cost, design flexibility and the opportunity to present products in an attractive way. However, compared to traditional packaging made of glass, metal, aluminum-laminated films or metallised films, transparent plastic packaging provides less protection against light and also in many cases, a poorer barrier against oxygen.
Transparent plastic packaging is increasingly replacing traditional food packaging made of glass and metal. The advantages of transparent plastic packaging are; its low weight, low cost, design flexibility and the opportunity to present products in an attractive way. However, compared to traditional packaging made of glass, metal, aluminum-laminated films or metallised films, transparent plastic packaging provides less protection against light and also in many cases, a poorer barrier against oxygen.
Transparent plastic packaging is increasingly replacing traditional food packaging made of glass and metal. The advantages of transparent plastic packaging are; its low weight, low cost, design flexibility and the opportunity to present products in an attractive way. However, compared to traditional packaging made of glass, metal, aluminum-laminated films or metallised films, transparent plastic packaging provides less protection against light and also in many cases, a poorer barrier against oxygen.
The effect of light in conjunction with oxygen can lead to rapid quality loss or spoiling of the packed food due to fat oxidation, vitamin and colour loss and adverse effects on the taste.
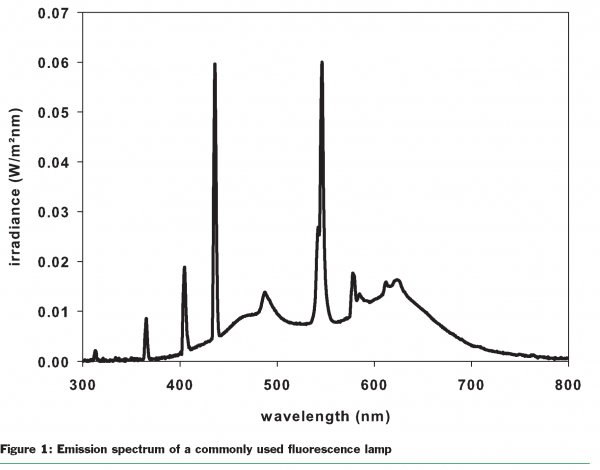
Food producers, packaging material manufacturers and retailers are very unclear about how retail lighting affects the quality and shelf-life of foods. The UV portion of retail lighting could play a particularly important role because of its high energy. Usually, the fluorescent lamps commonly used by retailers have characteristic bands at 315nm, 335nm and 365nm (Figure 1). Light of these wavelengths is high in energy. The quantum energy is given by the well-known equation:
E = h*v = h/c*λ (h = Planck’s constant, λ = wavelength)
This means, for example, that light having a wavelength of 380nm has a quantum energy of 319kJ/mol. This energy is high enough to extract protons from fatty acids and to form chemical radicals, which easily start autoxidation (molar proton binding energy in linoleic acid 270kJ, and in oleic acid 320kJ).

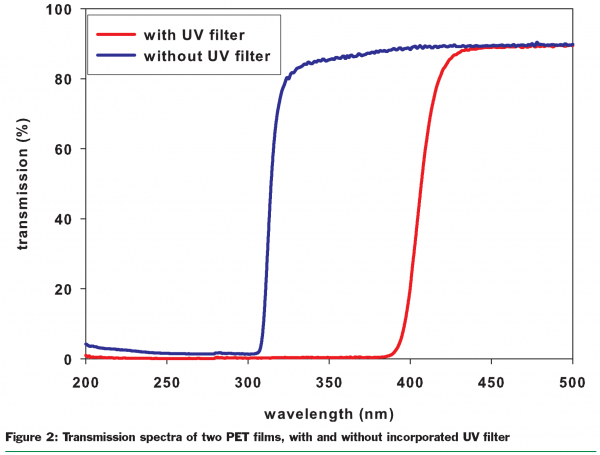
UV filters incorporated into packaging have the function of absorbing the major portion of the UV radiation that is incident on the packaging. Such filters protect the packed products (Rieblinger, 2003). Organic UV absorbers such as benzophenones and benzotriazoles are used for this. More recently, inorganic compounds such as titanium dioxide and zinc oxide have also been employed. An ideal filter should absorb light as close as possible to the UV/VIS transition region (380-400nm) in order to guarantee maximum protection for a fully transparent film (Danzl, 2005). Figure 2 demonstrates the reduced transmission between 300nm and 400nm by employing a UV filter in a PVC film. Nowadays, lamps used for illuminating supermarkets are sometimes already coated with UV filter layers. Nevertheless, UV filters incorporated into transparent packaging films have the advantage that they protect the packed goods against every external source of UV light, against retail lighting and against intense daylight.

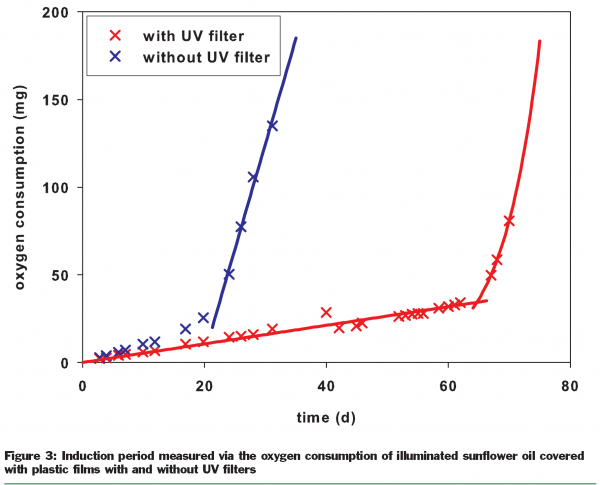
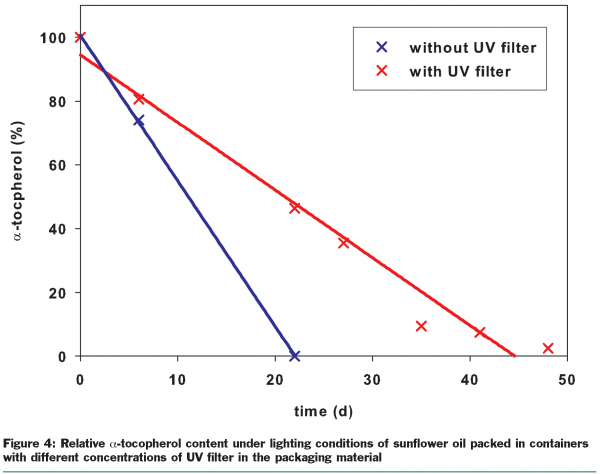
Typical UV damage to foods is often due to the auto-oxidation of fats, which is directly induced by energy input from light. UV filters provide a solution for this. The main focus is on refined plant oils, some animal fats and products such as snack products that are produced using plant oils. Other damage caused by UV light includes the discolouration of fresh meat, degradation of vitamins and the development of off-flavours. UV filters can also potentially provide protection here and so preserve the quality of the foods. For refined sunflower oil, it has been demonstrated that UV filters significantly prolong the shelf-life (Figure 3).There is prolongation of the induction period by about 200 per cent for refined sunflower oil, estimated via oxygen consumption. The end of the induction period is indicated by a marked increase in the oxidation rate and the beginning of rancidity, and therefore may be defined as the end of the shelf-life. However, it is not only the shelf-life which can be improved by the use of UV filters, but also the quality. This was shown by determining the concentration of tocopherols (vitamin E) in refined sunflower oil. After any period of storage within the shelf-life, the tocopherol concentration in the refined plant oil packed in containers with UV filters was higher than in the same oil packed in containers without UV filters (Figure 4). Such an increase in vitamin level means higher quality.


Besides their shielding effect towards lipids, UV filters may be of help in stabilising food flavour components. Carvon, the well-known flavour component of caraway, readily absorbs light between 230nm and 370nm due to its double-bond system and as a consequence undergoes rearrangement to carvon camphor which has a different flavour note. Therefore, UV filters are of interest in packaging systems for all foodstuffs that contain caraway as a predominant aromatic, for example, in plastic films for bread and in plastic or lacquered glass bottles for spirits (Refsgaard 1995).
It is very important to recognise here that it is not just high energy UV light that causes damage to foods but also lower energy light from the visible wavelength region. The reason for the latter is the ability of light to sensitise certain components of foods, and these are always coloured components (Sandmeier 1996; Ziegleder 2001). The best known examples are chlorophyll and riboflavin (vitamin B2) as well as all foods which contain small traces of these materials. Food components which typically contain chlorophyll are vegetables, herbs, spices and cold pressed plant oils. Food components which contain riboflavin are milk, dairy components, yeasts and bakery products, beer, eggs, fish and to a certain degree some cereals. It is therefore very important to define suitable application areas for UV filters in food packaging and to be aware of photosensitised reactions caused by visible light.
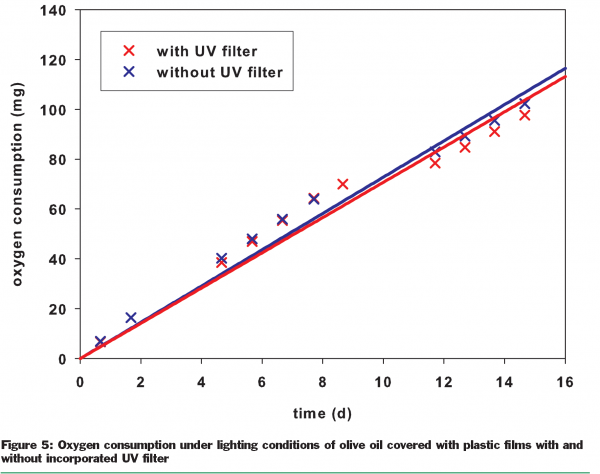
The sensitivity of plant oils to light is highly dependent on their origin and the processes used in their refinement. Cold pressed olive oil, due to the mild conditions under which the oil is recovered, still contains residues of chlorophyll and hence has the familiar green colour. It is absorption by the green chlorophyll that is the main reason for its sensitivity to visible light. Therefore, as shown by Figure 5, the presence of a UV filter in the packaging has no positive effect. In the presence of light and residual oxygen, the oxidation of cold pressed olive oil immediately begins without any induction period. Due to this lack of an induction period, such photosensitised oxidation is much faster than auto-oxidation. In addition, antioxidants such as tocopherols have no protective effect against photosensitised oxidation. In contrast to autoxidation, the photosensitised reaction does not occur via chemical radicals. Antioxidants cannot therefore intervene and catch radicals.

It is often the minor components of a food formulation which determine the specific sensitivity of a product (Sandmeier 1996). As snack products are made with refined vegetable oils, they are usually sensitive to UV light. Therefore it makes sense to use films containing UV filters to pack these products. However, if snacks contain special spices such as pepper or contain added yeast extract then these minor components will trigger photosensitivity. In this case, UV filters will either have no effect or only a partial effect as the deterioration is largely caused by visible light. Whenever herbs are added or spinach is used as a natural green colour, the effect will be photosensitivity to visible light. Rosemary is often used as a potent natural anti-oxidant. It is important, however, to be aware of the changed effect under visible light, because then rosemary switches to being a pro-oxidant due to its chlorophyll component. Within the broad field of dried soups, there are some products such as chicken soup which are sensitive to UV light because they contain highly sensitive unsaturated fatty acids. On the other hand, dry broccoli cream soup is typically sensitive to visible light because it contains the photosensitisers chlorophyll from broccoli and riboflavin from dairy ingredients. In order to identify useful applications for UV filters in food packaging it is therefore essential to pay attention to the specific formulations and to be especially aware of minor components such as natural pigments or extracts, herbs or spices. If photosensitive products are packed in transparent packaging, only a highly oxygen free atmosphere can provide good protection.
A well-known adverse effect of light is the discolouration of sliced sausage which often occurs even if the product is packed under vacuum (Andersen, 1992; Moller, 2002). Under retail conditions the exposed surface of the sausage may rapidly turn a greyish colour. As Figure 6 shows, the discolouration of the red colour component follows exponential kinetics. The fresh red colour of minced meat bleaches rapidly. The colourant in these cured meat products is the muscle pigment nitroso-myoglobin. We expected plastic films containing UV filters to retard the discolouration but the results were disappointing. Figure 6 shows the reduction of the red colour as a function of the storage time. Different packaging materials were tested (with and without UV filter) and tests were undertaken with different oxygen levels in the headspace. It is clear from Figure 6 that the UV filters had no effect. Nitroso-myoglobin, which gives sausage meat its red colour, develops during the mincing process and acts as a potent photosensitiser. It absorbs light from the visible region and transmits the energy to oxygen, which attains higher reactivity and starts the bleaching and oxidation processes. Although the UV filter in transparent packaging has a retarding effect on lipid oxidation, this positive effect has no practical relevance because the discolouration of the sliced sausage surface is much faster than lipid oxidation and rapidly leads to loss of product quality.
The rate of discolouration can only be reduced by markedly reducing the oxygen concentration (Figure 6). It must be recognised here that it is not easy to lower the oxygen concentration by vacuum or by using a modified atmosphere because oxygen may be dissolved in the product and will diffuse into the packaging headspace. A packaging free of oxygen was reached by a sequence of steps involving flushing the filled packaging with nitrogen followed by vacuum treatment. The sausage product in the oxygen-free packaging was stable to discolouration regardless of whether a UV filter was present or not.
Unlike the colour of sausages, the red colour of fresh meat is caused by oxymyoglobin. This substance needs the presence of oxygen to stabilise the red colour. For this reason, fresh meat is often packed in a modified atmosphere containing some percentage of oxygen. As the red colour can be destroyed by the effect of light energy, UV filters have a positive effect in this case. When plastic films containing UV filters were used to package the meat, the fresh red colour of meat was stable, whilst it turned brown when no UV filters were present in the plastic films (Bertelsen, 1986; Bertelsen, 1987; Martíneza, 2007).
A key aim of this project is to classify the specific sensitivities of various foods to light in the visible or UV region. This will allow suitable applications for UV filters in transparent packaging films to be defined. In many cases it is advantageous to combine packaging containing UV filters with modified atmospheres. By adopting this strategy, autoxidation as well as photosensitised reactions can be effectively retarded.
Acknowledgements
This research project was funded by the German Ministry of Economics and Technology (via AiF) and the FEI (Forschungskreis der Ernährungsindustrie e.V., Bonn). Project AiF 13858N. Films containing UV filters were provided by Dr. Deiringer, Klöckner Pentaplast.
References
- Sandmeier, D. (1996). Prooxidative Effekte von Gewürzen unter Lichteinfluß. Fett. Zeitschrift für Wissenschaft und Technologie der Fette, Öle und Wachse, 98, 199-202
- Rieblinger, K., Danzl, W. (2003). Vorsicht Licht! Verpackte Lebensmittel besser schützen durch Farb- und UV-Filterfolien. Lebensmitteltechnik, 35, 48-49
- Danzl, W., Ziegleder, G. (2005). UV-Filter in Lebensmittelverpackungen. Chancen und Grenzen für den Produktschutz. Verpackungsrundschau, 56, 63-66
- Ziegleder, G., Thron, M., Eichner, K. (2001). The influence of light of different wavelengths on chlorophyll-containing foods. Food Science and Technology, 34, 542-548
- Refsgaard, H. H. F., Nielsen, B. R., Skibsted, L. H. (1995). Sensory Importance and Mechanism of Potochemical Conversion of Carvone to Carvonecamphor in Ethanol-Water Mixtures. J. Agric. Food Chem., 43, 1177-1183
- Refsgaard, H. H. F., Brockhoff, P. M., Poll, L., Olsen, C. E., Rasmussen, M., Skibsted, L. H. (1995). Light-induced Sensory and Chemical Changes in Aquavit. Lebensm.-Wiss. u. -Technol., 28, 425-435
- Bertelsen, G., Boegh-Soerensen, L. (1986). The effect of lighting on colour of beef. In: Proceedings of II R-Meeting, Bristol, UK, 433–438
- Bertelsen, G., Skibsted, L.H. (1987). Photooxidation of oxymyoglobin. Wavelength dependence of quantum yields in relation to light discolouration of meat, Meat Science, 19, 243–251
- Martíneza, L., Cillaa, I., Beltrána, J. A., Roncalés, P. (2007). Effect of illumination on the display life of fresh pork sausages packaged in modified atmosphere. Influence of the addition of rosemary, ascorbic acid and black pepper. Meat Science, 75, 443-450
- Moller, J. K. S., Bertelsen, G., Skibsted, L. H. (2002). Photooxidation of nitrosomyoglobin at low oxygen pressure. Quantum yields and reaction stoechometries. Meat Science, 60, 421-425
- Andersen, H. J., Skibsted, L. H. (1992). Kinetics and Mechanism of Thermal Oxidation and Photooxidation of Nitrosomyoglobin in Aqueous Solution. J. Agric. Food Chem., 40, 1741-1750









